|
Today, I met with my mentor to discuss about my next experiment. The main purpose of the experiment is to examine the effects of different pH levels on the heartbeats of Daphnia Magna. We decided that it will be best to change the pH only slightly to best mimic the actual aquatic biomes. Although detergents or pesticides themselves could be very acidic or basic, once released in the freshwater, they could be diluted, leading to only a slight change in the pH of the water. Of course, an extremely low pH (acidic) or high pH (basic) would most likely kill the crustaceans. However, it is not so obvious what subtle pH changes would do to them. By changing the pH level slightly possibly by dilution, I would be able to find the pH range in which the D. Magna are able to survive.
The experiment will most likely take place as soon as I come back from winter break. I had hoped to finish it before I leave for winter break, but due to Revels rehearsals and final assessments, my mentor and I decided that it would be best to carry out the experiment after I come back. I would most likely arrange the arrival of the D. Magna to be the day after I come back so that I could start and finish the experiment the week I come back. Meanwhile, I am planning to do some more research. For instance, what is the pH range of freshwater biomes in its normal state and in its contaminated state? By knowing how extreme the pH is when the water is polluted, I would have a better idea of how much to dilute for my own experiment in order to most closely mimic a real life pH level.
1 Comment
In preparation for my main experiment next semester which is monitoring the effect of detergents on Daphnia Magna, I did some research on what happens to actual aquatic biomes when detergents show up in them.
Why Test Detergents? Any type of detergent, including the biodegradable ones, can have poisonous effects in all types of aquatic life. For instance, any type of detergent can destroy the mucus layers that protect fish from bacteria and parasites while causing severe damage to the gills. More specifically, surfactant detergents could significantly decrease the breeding ability of aquatic organisms. Detergent concentrations of 5 parts per million (ppm) will most likely kill fish eggs while 15 ppm could kill adult fish. Another problem that arises from detergents is lowering of the surface tension of the water. Thus, organic chemicals such as pesticides are much more easily absorbed by the fish. A detergent concentration of only 2 ppm, although not high enough to affect fish directly, can cause fish to absorb twice the amount of chemicals they normally absorbs. The chemicals in detergents also lead to natural disasters that eventually affect the aquatic organisms. For example, phosphates can lead to algal blooms that release toxins and deplete oxygen in the water. Once the algae decompose, they consume most of the oxygen, preventing aquatic organisms from absorbing it. Apart from phosphates, the sodium silicate solution and the surfactants are also major contributors to the toxicity of detergents. Why Use Daphnia Magna? Many laboratory experiments use Daphnia Magna as a indicator of toxicity and treatment efficacy of a potential resolution to an aquatic problem. The main reason is the crustacean's high sensitivity to toxic substances, short generation time, ability to multiply rapidly, ability to easily acclimatize in laboratory conditions, ability to culture in a small space, and allowance for measurement in a relatively short period. In several countries across the world, Daphnia Magna is used to monitor wastewater treatment systems, to determine permissible concentrations of pollutants, limits of impurity in water from natural effluents, and to determine the effectiveness of a sanitation method. Before I leave for winter break, I plan to complete another set of experiment following a similar method as the one for the first set, except measuring a different factor. I am meeting with my mentor tomorrow to decide on my experimental variable, but we have already talked about possible ideas in the past, one of which was varying the pH and examining how varied pH levels affect the Daphnia Magna. Last Wednesday, I received my Daphnia Magna kit and got straight to work. I set up my space in one of the Hunter classrooms to put my Daphnia Magna in. In order to control the light variable, I had fluorescent light turned on 24 hours and placed the tank containing Daphnia Magna under the light. I poured three liters of spring water into the tank, carefully submerged the container that the D. Magna was shipped in and tilted it so that the water flea could swim out into the larger tank. After, I poured another liter of spring water into the tank to fill it up to the brim. As the crustaceans were getting used to their new environment, I poured 1/8 teaspoon of green algae powder that came with the kit as food for the D. Magna. As time passed, the powder gradually submerged to the bottom, allowing the D. Magna to feed on it. That day, I struggled with the microscope, represented by the picture in my previous post, to view the D. Magna's heart beating. However, I simply was not able to focus the lens enough to distinguish the thinly outlined heart crammed next to all the other organs. As a result, I used a microscope that I have always used in my biology class last year since I knew how to handle it better. Soon after, this was what I saw:
Seven times and five time on Thursday and Friday, respectively, I measured the heart beats per minute of ten Daphnia Magna. I recorded each of the heart rates of the ten Daphnia Magna into my notebook. This is an example of what my data table looks like for every time I measure the heart rates of ten Daphnia Magna. As represented by the numbers, the heartbeats/min of the ten Daphnia Magna are more or less in the similar range. Later, I would average the ten data sets for easier comparison with the other averages that I will calculate for the other experiments I conducted at different times of the day. As I looked through the data that I recorded throughout the day over the course of two days, I noticed a subtle but interesting pattern. The overall heart rates of the Daphnia Magna were relatively higher in the morning and nights than in the middle of the day. On average, heart rates in the morning and night would average in the 400s or even 500s whereas those measured in the midday were in the 300s. As I collect more data, I hope to further testify the conclusion that I made from my first set of experiments by observing a similar pattern. My next steps are to conduct experiments following the same procedure except with a new focus. I have yet to decide on what I will compare the trend in the Daphnia Magna's heart rates to, but I hope my discoveries serve to provide a baseline data for the "real" experiment in which I will examine the effect of detergents on the Daphnia Magna. Here is a picture of me looking in to the microscope to count the heart beats. I am holding a timer with my left hand while tapping the table with my right hand to keep track of my counts.
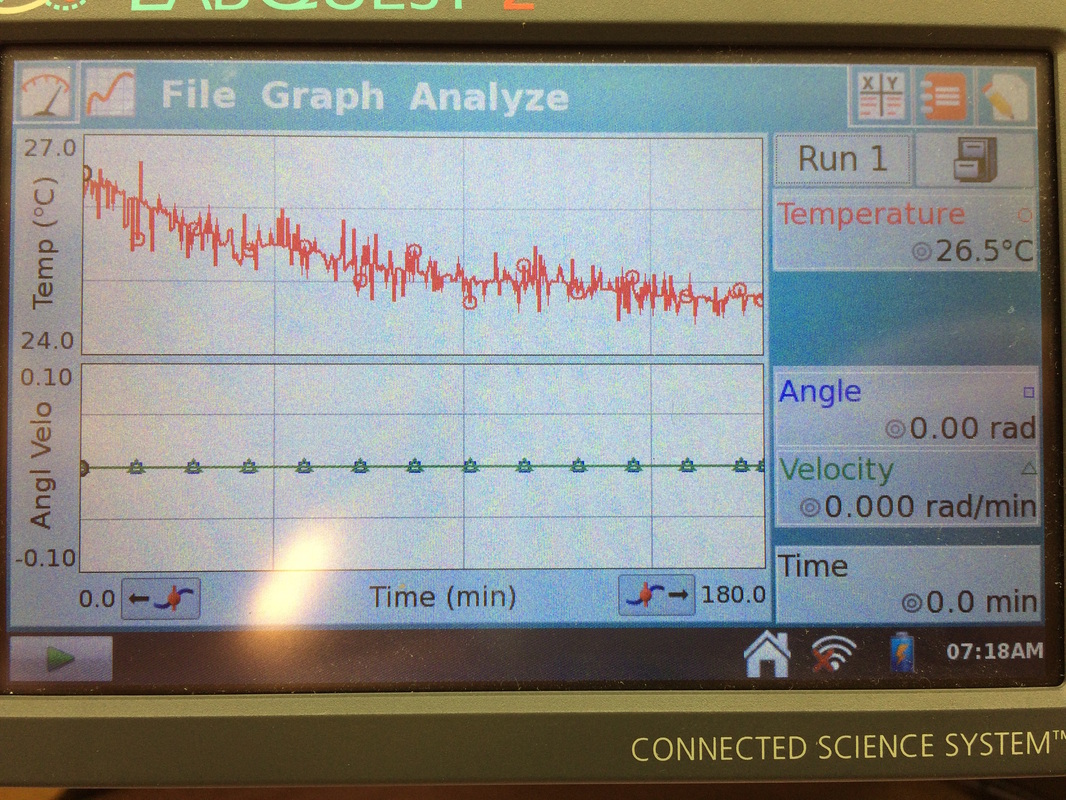
Next Wednesday, I am going to do my first set of experiment on examining the change in Daphnia Magna's heartbeats throughout the day. The purpose of this experiment is to investigate how the water flea's heartbeat changes at different times of the day and to use this data as the basis for my real experiment next semester. To recap, my goal of the project is to examine the acute cardiac effect of Daphnia Magna by incubating it into different diluted solutions of household detergents. For accurate data analysis, I have decided to do some pre experiments to learn about the different factors that could affect the crustacean's heart rates, the first one being different times throughout the day. Earlier this week, my mentor, Mr. Evans, ordered a Daphnia Magna kit containing a tank, Daphnia Magna, and food for the water flea, which is planned to arrive next Wednesday. In the mean time, I set up temperature probes in the STEAM room in the basement of Hunter, in the room right across the STEAM room, and in the two physics classrooms in order to find the best place to do my experiment. The optimum temperature for Daphnia Magna is 21 degrees Celsius or 69 degrees Fahrenheit. So far, the temperatures in the STEAM rooms are 25-26 degrees Celsius and 20-21 degrees Celsius in the physics classrooms. Since my experiment will last a maximum of three days due to the short life span of the Daphnia Magna, I hope I could use a small space in one of the physics classrooms for my experiment. This is a picture of the temperature probe that was left in the STEAM room to measure the room temperature for 3 hours. As shown on the right, the temperature reads 26.5 degrees Celsius, which is too high for the Daphnia Magna to survive in. This is a highly sophisticated microscope that Mr. Calos allowed me to use to look at the Daphnia Magna heart with. The blue camera on the left is a camera that is attached to the scope. I could use it to take pictures of what I see or even take videos. It would be really cool to take videos of the Daphnia Magna's heart beating!
I look forward to starting my experiment this coming Wednesday and learning about the pattern of changes that the Daphnia Magna's heart goes through. Below is a list of the tasks I hope to accomplish in each month throughout the year.
September: -Decide on the sample size and age group for initial experiment -Learn about how to take care of "D. magna" -Decide on factors that I will be focusing on in my initial experiment (ex. heart rate during different times of the day) October: -Order a kit of "D. magna" -Observe and record findings for 3-4 days -Repeat several times with different age groups (adults, juveniles, neonates) and compare results November: -Decide on which detergents to test in the spring -Come up with an experimental procedure for the spring experiment December: -Plan on specific details of experiment (volume of detergent, number of detergents testing, when to start the experiment, age group of "D. magna" that will be used) January: -Gather materials for experiment (detergents, "D. magna", lab equipments) -Start experiment as soon as possible -Gather first set of data February: -Perform multiple trials of experiment (possibly with different detergents or different age groups if time allows) March: -Analyze results into graphs -Start making presentation slides April: -Finish making power point -Practice presenting May: -Continue to finalize power point and practice presenting -Present during reunion weekend Hello, my name is Alice Kim, and I am a senior at Emma Willard School. I have been in the STEM program at Emma since my freshmen year and in the STEAM Signature program since my junior year. I am highly interested in pursuing my college major in environmental engineering, which is what my senior research project is based on. Last year, I worked on improving the happy light through summer scents in which I successfully completed experiments to prove my hypothesis and presented at the end of the year. This year, I want to focus my project mainly on environmental issues and examine the acute cardiac effect of detergents on "Daphnia magna". To give a little overview of how I chose my research topic, I participated in the Greater Capital Region Science and Engineering Fair during the spring break of my junior year with a project in which I examined the acute cardiac effect of Roundup on "Daphnia magna". Roundup is a company that produces "eco-friendly" weed killers. However, there have been many claims that refute this statement, so I engaged in an extensive research in which I carried out experiments on a water flea called the "Daphnia magna" to scientifically prove the claims. Following this successful research project, I wanted to expand on my research with the water flea in a different direction. Considering a fair number of science experiments use "D. magna" due to its transparent body that allows scientists to easily measure its number of heartbeats under a set time, measuring the acute cardiac effect would be an effective way to test my new hypothesis: detergents negatively impact the marine life.
|
AuthorHello! My name is Alice Kim, and I am a senior at Emma Willard. I am interested in learning about environmental issues. ArchivesCategories |
||||||



 RSS Feed
RSS Feed
